Weseemed Clinical Experience Sharing Initiative

Eligibility
- Open to licensed healthcare professionals currently using Weseemed--RP11A or SoloSure (SP11A) in real clinical practice.
Distributors, agents, and non-clinical personnel are not eligible.
How to Participate
To participate, eligible participants must:
- Follow Weseemed on LinkedIn.
- Publish at least one photo or one video based on real clinical use on LinkedIn or another publicly visible professional platform.
- Include #Weseemed in the post, and tag or mention Weseemed where available.
- Only publicly visible posts are eligible.Private, deleted, or restricted-access posts will not be considered valid submissions.
Types of Shareable Content
Participants may publish photo or video content on professional platforms such as LinkedIn, including:
- Clinical application scenarios
- Procedure demonstrations and practical experience
- Educational or academic exchange segments
- Product feedback and improvement suggestions
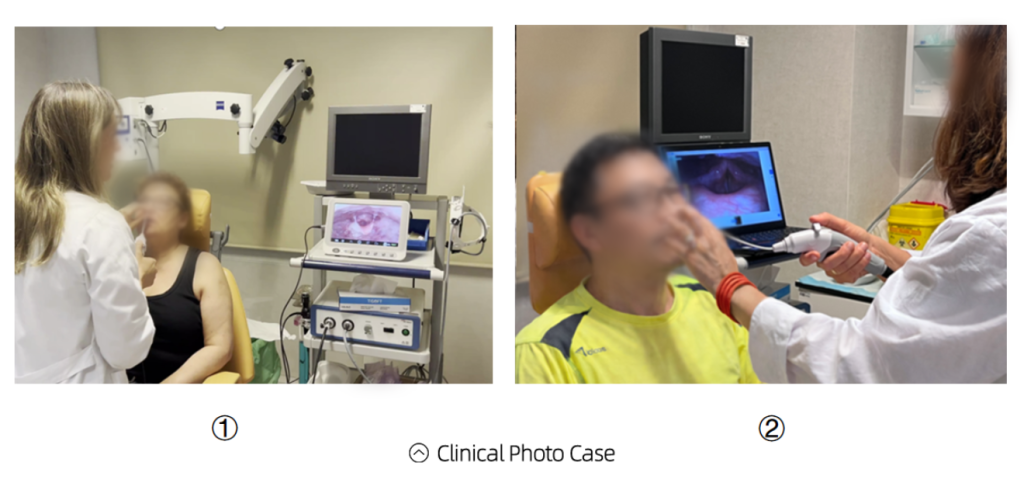
Content Requirements
✔ Real clinical usage scenario
✔ Clear and stable visuals
✔ No patient privacy information
✔ No promotional or exaggerated language
✔ Product serial number visible
✔ One submission per participant
- Each participant, each social media account, and each product serial number may be used only once per campaign. Duplicate, repeated, or substantially similar submissions may be disqualified.
Submission for Review
- After publishing the post, participants must send the following information to marketing@weseemed.com for review:
- Doctor’s full name
- LinkedIn profile name
- Product serial number
- Post link
- This information is required for identity and eligibility verification by the Weseemed team.
Product Collection
Approved participants may collect the product at the Weseemed booth during the exhibition, subject to availability.
Participants may be required to present proof of identity, post link, and product serial number.Unclaimed product support after the exhibition will be deemed forfeited unless otherwise confirmed by Weseemed.
- In case of force majeure (e.g., exhibition postponement) or personal circumstances preventing attendance, eligibility may be carried over to a future event, subject to WESEE’s confirmation.
Compliance Statement
- Weseemed will review submissions based on authenticity, professionalism, compliance, and completeness.
- Weseemed reserves the right to reject content that does not meet campaign requirements.
- Review decisions are final.
- Approved participants will be notified individually.
- Weseemed reserves final interpretation rights within applicable laws/regulations.
- Only posts published during the official campaign period are eligible.
- The campaign is valid until May 30, 2026, or until all available units have been claimed, whichever comes first.
- ©2026 All rights reserved by Zhongshan Wesee Meditech Co., Ltd.
Privacy and Data Protection
- Participants may be required to provide information such as name, professional credentials, product serial number, and content links.
- Such information will be used only for participation review and communication, and not for marketing purposes.
- WESEE will process and protect such data in accordance with applicable laws.
Compliance Statement
This initiative is governed by the laws of the People’s Republic of China. Any disputes arising from this initiative shall first be resolved through friendly consultation; failing which, they shall be submitted to a competent People’s Court in China.







